Quantitative PCR (qPCR), also called real-time PCR, is a molecular biology technique that enables the quantification of nucleic acid sequences in real time during the amplification process. This method combines the principles of standard PCR with fluorescence-based detection, allowing researchers to measure the accumulation of DNA with high sensitivity and specificity throughout the PCR cycles.[1][2][3][4][5]
Key Steps in qPCR Methodology
- qPCR begins with the preparation of a reaction mix containing DNA polymerase, primers targeting the region of interest, dNTPs, and a fluorescent reporter—either an intercalating dye (like SYBR Green) or a sequence-specific probe (like TaqMan).[2][4][1]
- For gene expression studies, RNA is first extracted and reverse-transcribed into complementary DNA (cDNA); DNA samples can be used directly for DNA quantification.[3][5]
- The reaction is placed in a specialized thermal cycler equipped to detect fluorescence. Typical amplification consists of 35–40 PCR cycles, each with:
- During each cycle, the reporter emits fluorescence proportional to the amount of DNA produced. The instrument records these signals after each cycle, enabling quantification.[4][1]
- The cycle threshold (Ct or Cq) value—when the fluorescence exceeds background—is used for quantification. Lower Cq values indicate greater starting nucleic acid amounts.[2][4]
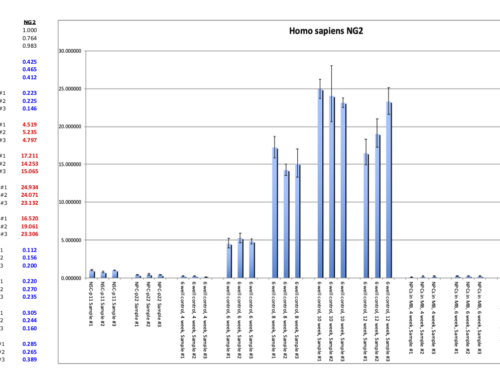
Quantification and Analysis
- Absolute quantification uses a standard curve of known concentrations for calibration.
- Relative quantification (especially in gene expression) compares Ct values between test samples and reference genes (housekeeping genes) using methods like ΔΔCt:
RQ = 2(ΔΔCt)
where
ΔΔCt = ΔCt (experimental sample) – ΔCt (control sample) for both test and control samples.[2]
Controls and Best Practices
- Negative (no template) controls ensure no contamination.
- Positive controls confirm assay validity.
- Endogenous (housekeeping gene) controls enable accurate normalization of gene expression.[2]
qPCR offers rapid, precise, and contamination-resistant quantification of nucleic acids, making it invaluable for applications such as gene expression profiling, pathogen detection, and genetic mutation analysis.[5][3][4]
ARQ Genetics has many years of experience in all aspects of qPCR projects, including nucleic acid extraction, design of both primer and probe-based assays, qPCR services and even data presentation. We’re here to support your ongoing research efforts so contact us today!
- https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/genomics/qpcr/how-qpcr-works
- https://www.the-scientist.com/insights-into-qpcr-protocol-detection-methods-and-analysis-71478
- https://www.thermofisher.com/us/en/home/life-science/pcr/real-time-pcr/real-time-pcr-learning-center/gene-expression-analysis-real-time-pcr-information/introduction-gene-expression.html
- https://biosistemika.com/blog/qpcr-technology-basics/
- https://www.bio-rad.com/en-us/applications-technologies/what-real-time-pcr-qpcr?ID=LUSO4W8UU
- https://www.youtube.com/watch?v=iu4s3Hbc_bw
- https://pubmed.ncbi.nlm.nih.gov/24011054/